Beyond lithium - Annual Report 2018
8 April, 2019

New batteries: three approaches
To build more sustainable batteries, should we look to different materials?
In today’s tech-hungry world, lithium batteries are ubiquitous. Everything from your mobile phone to the neighbour’s electric car rely on the metal, and it’s easy to see why. Lithium-ion (Li-ion) batteries pack a punch, storing more energy than any other battery of equivalent size, and delivering power to where it’s needed, quickly and efficiently.
But they’re far from perfect. For a start, lithium batteries come with a significant environmental footprint – extracting a tonne of lithium requires more than two thousand tonnes of water – and at the end of its life, a Li-ion battery is usually dumped in a landfill or incinerator, thanks to a lack of practical recycling routes. In operation, lithium batteries can overheat, and as news headlines have reported since 2013, they can combust, or even explode, when damaged. And finally, the materials that go into making Li-ion batteries – namely lithium and cobalt – are not Earth- abundant. Eventually, we will run out of them.
With demand for reliable batteries growing exponentially, what can we do? According to MacDiarmid Institute Principal Investigator, Professor Thomas Nann from Victoria University of Wellington, the answer is to look elsewhere on the Periodic Table of Elements. Specifically, at a very familiar metal – aluminium. “We found that aluminium’s potential energy density (a measure of how much energy a material can store) comes very, very close to lithium’s, but it has the added bonus of being the third most abundant element in the Earth’s crust,” he says. Professor Nann and his team took that as a starting point, and set out to design a new aluminium-ion battery.
They wanted to stick with a proven battery architecture – namely, two electrodes separated by an electrolyte – but the chemistry needed a shakeup.
Their first target was the electrolyte material itself, which, because ions move through it, is vital to the battery’s operation.
It is usually made from an expensive cocktail of compounds, so in search of a cheaper alternative, Professor Nann and MacDiarmid Institute-funded PhD student, Nicolò Canever, looked to the mining industry. “There’s a compound called acetemide that’s already used to extract aluminium ions from solution, so we decided it would be the basis for our electrolyte,” Professor Naan explains, “It can also be produced by bacteria, so is incredibly inexpensive.” Their final compound worked well, and even allowed for ultrafast charging.
While that work was recently published in a Royal Society of Chemistry journal, the team’s design of a new electrode material has taken a different route. “It is so transformative and so surprising that we knew we had to patent it,” Professor Nann says. He credits his PhD student Shalini Divya with taking up the challenge of starting from scratch, rather than simply improving on what had gone before, “She found a material that outperforms everything that’s been published to date”.
A key step in the patenting process is to prove that their lab-produced batteries could be manufactured in a commercial facility, so Professor Nann and Ms Divya recently travelled to Germany’s Fraunhofer Institute to do just that. The trip, funded by the MacDiarmid Instiute, was a huge success. They arrived back to New Zealand with twenty of their novel aluminium batteries, which Professor Nann says are “… approaching the performance of lithium ion batteries currently on the market.” Best of all, they could be produced with only very minor changes to existing processes, “…which is a key consideration for potential investors or manufacturers.”
Professor Nann and his team are continuing to develop their patent and their battery at a time when the need for sustainable energy storage has never been more urgent. “As we transform into an energy landscape dominated by renewables, the problem is not getting hold of energy – after all, if we covered 250 by 250 square kilometres of the Australian outback in commercial solar panels, we’d generate all the energy our entire planet needs,” he explains. “This isn’t as much area as it initially seems – rows of solar panels alongside eexisting highways could make a big impact.”
Professor Nann is driven by adding value through his science.
“The most pressing issue is matching supply and demand, and that’s where our work comes in. I want to make a difference; to have a real impact on energy storage in New Zealand and around the world.”
Can we skip the solar cell altogether?
Another battery project connects Professor Nann with his MacDiarmid Institute collaborators and University of Canterbury researchers, materials engineer Professor Maan Alkaisi, chemist Associate Professor Vladimir Golovko, and chemical engineer Associate Professor Aaron Marshall.
Associate Professor Marshall says the project is funded under MBIE Smart Ideas, and involves looking at new smart materials that can convert sunlight directly into stored battery energy, without making electricity in the process (i.e. skipping the whole solar cell creation of electricity step).
“Currently the challenge is to find a material which absorbs the sunlight and catalyses the charging reaction directly inside the battery”
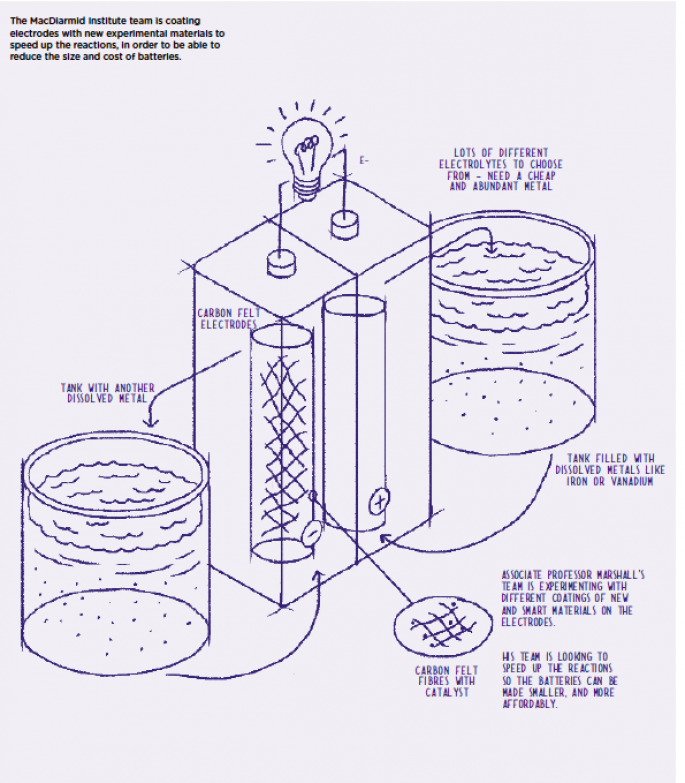
Shrinking monster-sized flow batteries
Associate Professor Marshall and his MacDiarmid Institute collaborator University of Canterbury chemist Professor Alison Downard are also working to speed up (and shrink) redox flow batteries. In a redox flow battery, the ‘energy’ is stored in chemicals which sit in (usually big) tanks separate to the battery itself. When energy is needed, the chemicals are pumped through the battery and through the porous electrodes.
 “The concept it a bit like filling your car’s fuel tank with petrol – you could then leave the car for a year and it would still have a full tank of gas and be ready to drive when you needed it.”
“The concept it a bit like filling your car’s fuel tank with petrol – you could then leave the car for a year and it would still have a full tank of gas and be ready to drive when you needed it.”
And he says the chemicals (usually a dissolved metal species like iron, vanadium or bromine) are relatively abundant and therefore relatively cheap over the lifetime of the battery. But the batteries are currently slow.
“Slow reactions means the battery requires big electrodes. And if the electrodes are large, the rest of the battery has to be large as well, and the whole thing ends up being expensive.”
So the MacDiarmid Institute team at the University of Canterbury is looking at ways to coat the electrodes with new materials to speed up the reaction – so they can reduce the size of the electrodes. They’re using the vanadium electrolyte system to test changes against; vanadium is well studied in redox flow batteries and therefore a useful standard system with which to trial improvements. If they can improve the reaction rates by two to three times without losing efficiency, these new electrodes would make flow batteries very competitive.
Associate Professor Marshall says he and Professor Downard share supervision of a PhD student Leatham Landon-Lane who is working full-time on this project.
“MacDiarmid funds supported the student to travel to Australia this year to study the surface chemistry of their electrodes at La Trobe University’s Centre for Materials and Surface Science.”


